Snapsil’s newer focus on the healthcare market leverages the pack’s hygienic and easy-open features for powders, liquids, solid dose tablets, and medical devices.
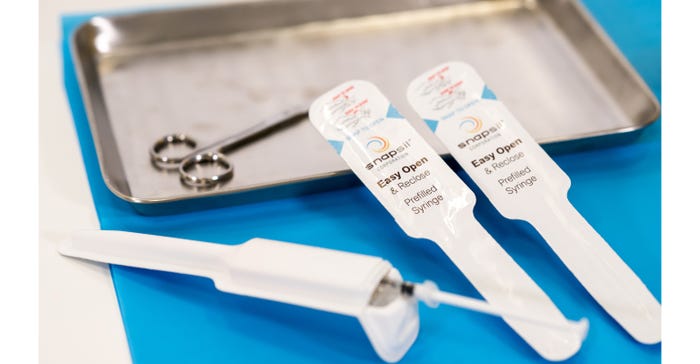
Sealed medical packages maintain their integrity until opened, protecting devices such as pre-filled syringes.Photo supplied by Snapsil
When Packaging Digest first reported on Snapsil single-use packages in 2014, target markets included food, personal care, healthcare, and household products. And its first commercial product was for tomato sauce. But Snapsil packs are particularly suitable for medical products because of their hygienic design, which is made even more useful because of the easy, one-hand opening feature.
Neil Cashman, Snapsil CEO, talks about global trends that support the package’s benefits for healthcare applications (also see slideshow for examples of packages holding different types of products).
When did you decide to focus on the medical market?
Cashman: We introduced Snapsil medical applications to customers in 2019. This was in response to what we see as a growing need for “easier to use” packaging to assist with patient treatment adherence and an aging population. Here’s how the package addresses both these needs:
1. The packaging contributes to patient non adherence. The correct and timely use of medication determines its therapeutic effect. Yet physical constraints — such as poor handgrip strength, loss of fine motor skills, and impaired vision — negatively impact the taking of medicines. In developed countries up to 50% of patients fail to adhere to treatment, resulting in an estimated $600 billion in lost sales annually.
Studies have shown that many people, especially elderly, experience difficulties when attempting to use medication packaging formats contributing to non-adherence. Medication packaging needs to become more focused on the patient and facilitate human interaction for a future that will see an aging population and increasing self-care.
2. Aging population is a global mega trend. In 2014, nearly one billion people were registered as late lifers — senior adults aged more than 60 years. By 2030, it is expected that this number will reach 1.5 billion people, representing 18% of the total population, and by 2050, the number will reach 2.1 billion. Older people are high users of medications. In meeting the medical needs of an aging population, there are challenges to overcome not only for drug development but also how these drugs are provided through packaging to senior patients. Poor packaging causes inconvenience, non-adherence, and, in some cases, injury.
In contrast, patient-friendly packaging can improve dosing, treatment adherence, patient safety, and mobility.
Numerous studies have identified the challenges elderly patients encounter when using existing pharmaceutical packaging formats and, as this mega trend (aging) continues, the need for patient-friendly packaging will become greater.
What percentage of your business does medical represent and why?
Cashman: We anticipate medical will become the largest area of the Snapsil business over the next few years as medical customer projects progress further.
We are excited by medical customers’ interest in Snapsil technology across the breadth of medical product applications — powders, liquids, solid dose tablets, and medical devices. We believe the underlying need for “easier to use” packaging is heightened in medical to assist patient treatment adherence especially amongst the elderly who experience reduced dexterity or patients who may be suffering ailments that may hinder their ability to open packaging.
Are the Snapsil products for medical applications made any differently? If so, how and why?
Cashman: Snapsil’s technology is applicable to packaging utilizing thermoplastic production processes used in both the medical and consumer goods industries. Snapsil cooperates globally with Multivac, who can assist customers with machine solutions where required.
Who are your U.S. material and production partners for medical?
Cashman: In the US, we currently cooperate with Amcor, who can assist customers with material solutions.
Due to the regulations in medical, Snapsil has not sought to limit customers production options by appointing a nominated chief medical officer (CMO). We find medical customers consider in-house production or may prefer to use their existing and qualified CMO.
What materials are typically used for medical packages and why those?
Cashman: Snapsil technology has both commercialized and trialed positively in a range of thermoformable polymer-based materials. We choose those materials to provide a range of advantages including ease in manufacturing and supply chain, speed to market through using commercially available options, and the ability to incorporate required material properties like barrier, light transmission, processability, and more.
For medical and pharmaceutical packages, Snapsil is also currently developing design options through the use of materials available from specific sites currently supplying the pharmaceutical markets.
About the Author(s)
You May Also Like




