Medical Packaging
Healthcare-GettyImages-843421738-ftd.jpg
Pharma & Medical
Healthcare Packaging News and Top IssuesHealthcare Packaging News and Top Issues
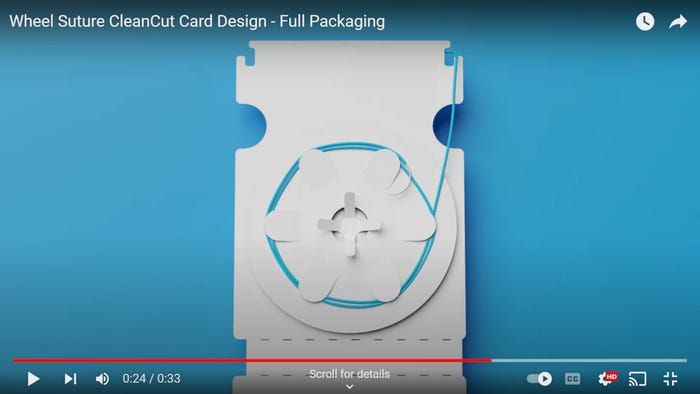
Medical packaging becomes work of art, environmentally friendly plastics, IoPP’s Mid-South Chapter meeting, more.
Sign up for the Packaging Digest News & Insights newsletter.

















.png?width=300&auto=webp&quality=80&disable=upscale)




.jpg_(1).png?width=300&auto=webp&quality=80&disable=upscale)






.png?width=300&auto=webp&quality=80&disable=upscale)







