4 options for in-line 100% seal inspection
March 22, 2017

For sterile medical devices, it’s critical that the seal on the package be completely intact to maintain product sterility. Seal defects can include voids, wrinkles, dust, random particles, or hair—all of which threaten the integrity of the seal. Inspecting seals after they’ve been formed has traditionally been a manual, visual process, done on a sample basis. Destructive package integrity testing may also be used, but this results in the package being discarded and the contents being rehandled on the production line for repackaging.
When producing Class III medical devices, a sampling strategy leaves room for failures to escape into the field, since the samples qualify an entire lot, rather than a specific item. Manual inspection can also be unreliable: humans get tired, get distracted, or get confused with borderline defects. As a result, lots that should be reworked might end up being shipped instead.
But technology is changing. It’s now possible to ensure the quality of the outgoing product by integrating inspection into the production line for 100% non-destructive inspection. Using carefully constructed and calibrated machine vision algorithms gives users more reliable inspection results than can be obtained manually, and individual failures can be caught even within high-quality lots, without slowing production.
A range of inspection technologies are available for integration into the production line. Suitability depends upon the application—in particular, on the type of packaging and the size of defect that needs to be resolved.
Following are four 100% non-destructive technologies that could be used for inspecting seal integrity:
Visible-light imaging: This comes in two forms: area imaging, which uses a conventional camera, and line scanning, which uses a one-dimensional array of charge-coupled device (CCD) light sensors. Both of these approaches rely on at least one of the packaging materials to be transparent. *Area imaging uses one or more cameras to capture pictures from all or a portion of the area to be inspected. Multiple images from one camera or images from multiple cameras may be stitched together to form a larger, more complete image. A single area image captures more information than a single line-scan image, and it can provide high resolution—down to 11 µm. It can be harder to light a full area uniformly; if that becomes an unresolvable problem, line scanning may be a better solution. *Line scanning records a single line of visual light; multiple lines are stitched together to form a larger image if needed. It requires less processing and is easier to illuminate than area imaging, and so it can be a better choice when area imaging becomes too complex or hard to light. Resolution is on the order of 500 µm.
Thermal and Infrared lighting: Heat signatures can be detected in the infrared range. Thermal sensing relies on the passive emission of infrared radiation from the package. One benefit of in-line inspection is that the amount of heat being radiated may be higher right after sealing, providing a stronger signal. Infrared imaging is similar, except that the scene is lit with infrared light and the reflections are imaged. Resolution for both technologies will depend on the image sensor. This approach is effective with aluminum, PET, polyethylene, and paper packages.
Opposing lasers look for variations in thickness by shining two directly opposing laser beams on the package seal. This allows the thickness of the package to be measured and for deviations to be flagged. This can be done on any material, with resolution at the micron level.
Ultrasonic approaches resemble the laser methodology, but, instead of measuring light reflections, they measure transmission of the transmitted ultrasound signal through the package. If the seal is intact, most of the signal makes it through to the other side. Gaps and particles, however, will cause significant internal reflections, reducing the transmitted signal. Resolution is poorer at 2 mm. It’s effective on Tyvek, paper, foil, film, aluminum, and plastic packages.
These technologies are listed together in the below table for comparison:
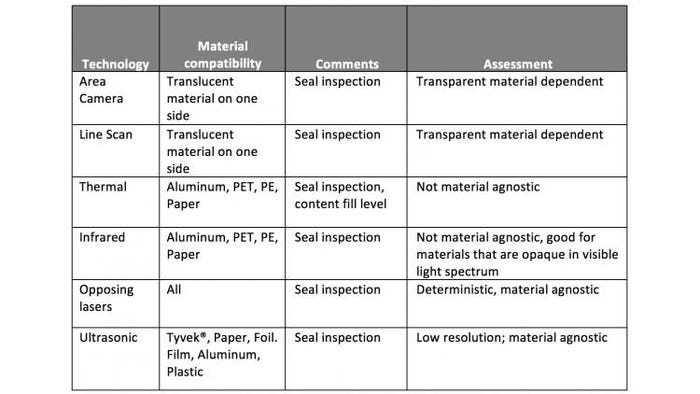
Above: Comparison of inspection technologies.
Below: The same Class III medical device seal captured with an area camera using an image algorithm.

Any of these technologies can be included in an automated system that implements 100% in-line inspection with no loss of throughput. But, when planning an integration project, it’s important to understand the material implications and the required resolution early so that the appropriate inspection approach can be selected and successfully integrated.
Bhaskar Ramakrishnan is Technical Sales Engineer for DWFritz Automation Inc. He may be reached at [email protected].
Be sure to visit DWFritz Automation at ATX East 2017 at Booth #2305 in New York City June 13-15.
About the Author(s)
You May Also Like


