3 steps for designing the ideal medical device packaging system
January 24, 2017

Medical device packaging design is an integral part of delivering your device to market safely and securely, with the sterile barrier intact. Yet this critical process is often given little attention in the overall product development process. Most single-use sterile medical devices can be opened with a high degree of confidence that the product has maintained sterility throughout the product lifecycle, yet getting to this point can prove troublesome. Here are three steps to consider when designing your ideal medical device packaging system.
1. Start packaging design early in the product development process.
Thoroughly defining the requirements of a packaging system early in the product development process sets the stage for long-term success. Prequalifying those requirements prior to full package system validation will save time and money in the long-run. Preliminary evaluation of product-package compatibility allows for early detection of design or manufacturing problems and affords time for revisions prior to full validation. Identifying possible packaging system failures and addressing them proactively during early design phases greatly increases the chance of successful validation and patient safety. When this step is skipped or left to the final hour, short-cuts often utilized to save time and money can increase the risk of noncompliance with regulations. Instead, during this process different sterile barrier systems can be evaluated and manufacturing methods evaluated to best meet the needs and characteristics of a particular device.
Test failures are not an uncommon occurrence and must be taken into account during the development process. Failure to plan is planning to fail. By starting this process early in product development, these contingencies can be addressed early, and unwelcomed delays and added costs can be avoided.
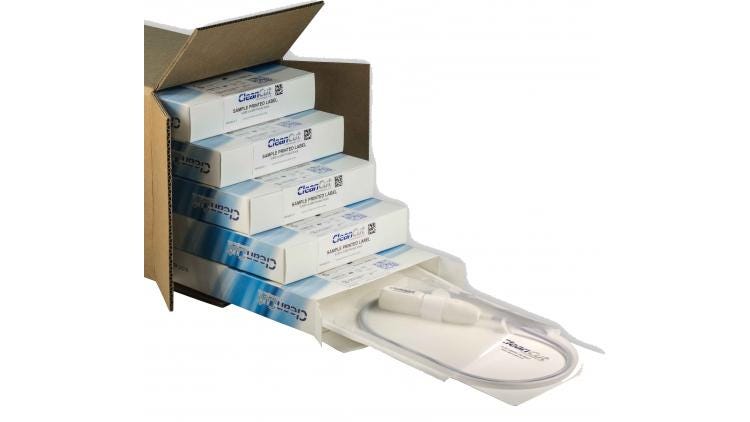
2. Properly size packaging components.
Loss of sterile integrity, one of the most common types of packaging failure, is often the result of improperly sized packaging elements in relation to the device. These packaging elements include the backer card or tray to retain the device, the pouch or lid, the shelf carton, and finally the corrugated master shipping container. If any one of these elements is improperly designed or sized in relation to the other, unnecessary movement will likely create complications during transportation and result in failure of your sterile barrier system.
For example, if the backer card is too large, constant pressure will be placed on the pouch seals. If it is too small, excessive motion within the pouch will create friction and abrasive forces. A pouch or tray lid within a shelf box that is too large will need to be folded or creased; one that is too small will be subject to repeated contact against the inner box, all of which cause stress and fatigue leading to pinholes and pouch failure.
Individual shelf cartons need to fit snugly around their contents to prevent movement during transportation, while the master shipping container must maintain sufficient rigidity and wall strength to protect the inner cartons consistent with the distribution environment of the device. All of these elements must be customized to the device, and they must work in tandem to reduce the risk of failure and successfully deliver your device to market.

3. Utilize a robust testing protocol.
Packaging concerns should be addressed from the very first step of the project to quickly identify unforeseen contingencies in design. Short-cuts simply cannot be taken during this critical testing process. ISO 11607: Packaging for Terminally Sterilized Medical Devices, published in 2006, is the principal reference guide for medical device packaging and includes information on testing requirements. ASTM and ISTA Series distribution testing offer a set of specific test methods for compliance with ISO 11607.
For guidance related to a specific medical device packaging system, collaboration between a project team and an ISO 17025 accredited laboratory is necessary to ensure the proper test methods are utilized. Establishing a protocol prior to testing complete with clearly understood pass/fail criteria is paramount to identify problem areas in packaging design. A robust protocol would include scope, responsibilities, appropriate sample size, production equipment, parameters, and acceptance criteria, to name a few.
Your Ideal Packaging System
In the competitive landscape of the current medical device marketplace, almost as important as the medical device is the package it arrives in. Packaging today has evolved from an afterthought to an integral part of the device user’s experience. In many cases, the packaging system is designed to be as unique as the device itself. Much needs to be considered in the process. The packaging system must maintain the sterile barrier system, protect the device while in transit and use, and successfully communicate the device maker’s branding and value proposition. When this process is considered early with great intent and purpose, mistakes can be avoided, costs can be saved, and the ideal packaging system can be created.

Brian Whalen (above) is director of sales & marketing for CleanCut Technologies LLC (www.cleancuttek.com). The company will be exhibiting at Booth #1361 at MD&M West in Anaheim, CA, February 7-9, 2017.
All images in this article are provided by CleanCut Technologies.
About the Author(s)
You May Also Like


