New blister-packaging concept uses pouches to provide product protection and child-resistance
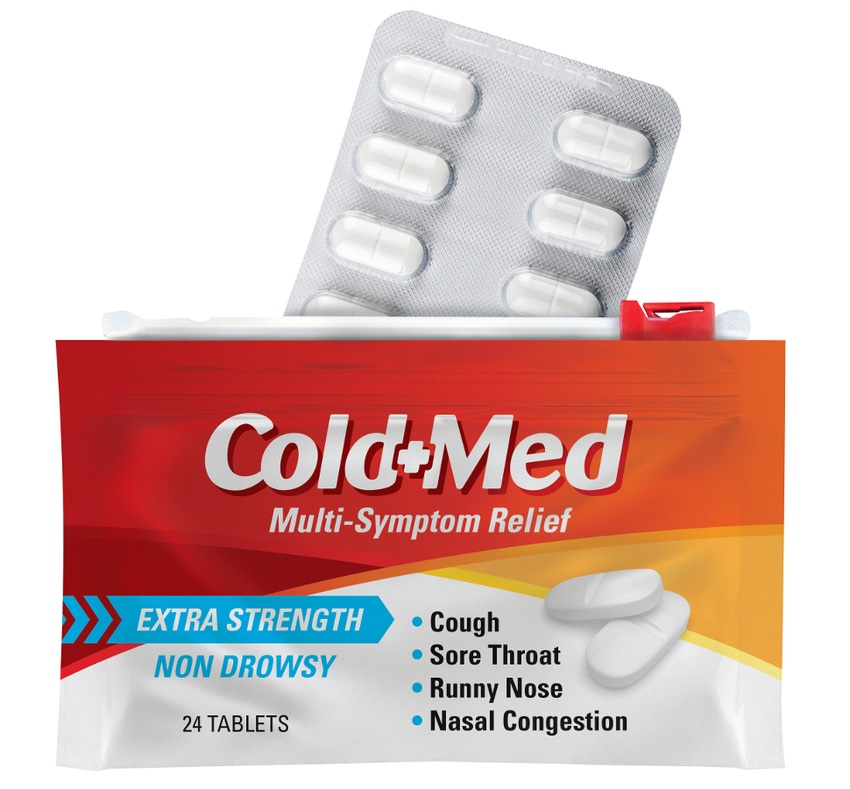
A uniquely functional pharmaceutical packaging concept, developed collaboratively by Bemis Healthcare Packaging and Presto Products Co., uses flexible packaging instead of paperboard cartons to protect blister packs.
The centerpiece of the packaging design is a pouch that provides both child-resistance (CR) and barrier protection for prescription and over-the-counter drugs. In addition, the design also offers merchandising flexibility for retailers and pharmacies.
The pouch features Presto’s Child-Guard CR track and slider, a closure designed to be difficult for kids to open but easy and intuitive for seniors.
According to Presto, the ergonomically designed closure has a Drug Master File listing, meaning it is FDA-compliant for use in pharmaceutical packaging. The reclosable slider meets the Poison Prevention Packaging Act’s CR requirements, as well.
The package also provides robust product protection. Bemis Healthcare Packaging converts the pouches using multilayer laminations that provide high oxygen and moisture barrier; depending on the product, the film may contain a layer of aluminum foil. Consequently, the primary blister pack can be made from cost-effective non-barrier materials—the pouch does the heavy lifting vis-à-vis product protection.
Georgia Mohr, marketing director-pharmaceuticals at Bemis Healthcare Packaging, answers a few questions about the package.
From what materials is the pouch film made?
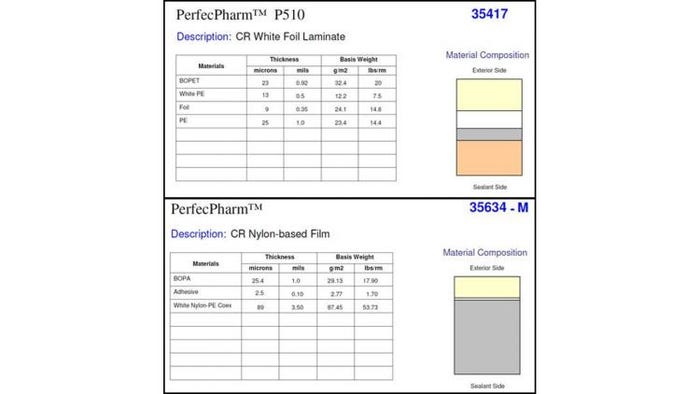
Mohr: We have a multi-layer lamination of 92-gauge PET/35-ga foil/PE (PerfecPharm P510). This lamination offers high moisture and oxygen barrier. The 92-ga PET is often used in foil laminations that will be used in CR packaging applications.
We also produced a pouch from a film/film lamination (PerfecPharm 35634-M). This non-foil lamination provides high puncture resistance suitable for CR packaging applications. (See charts below.)
How many layers are in the films?
Mohr: The PerfecPharm P510 is a three-ply structure with foil in the core. The 35634-M is a two-ply structure.
What size(s) does the package come in?
Mohr: The pouches are custom sized to meet the needs of the application.
How is the pouch printed?
Mohr: Either surface or reverse flexo print.
The pouch can accommodate a peg hole. Would this package normally be merchandised in a hanging display?
Mohr: The pouches can be created as stand-up or flat pouches, with the option to add a peg hole. So the pouch can be displayed on a peg. It gives our customer the flexibility to determine how they would like to go to market with the package.
Is the blister pack inside the pouch a standard packaging component?
Mohr: The blister inside the pouch is a common blister size.
Can the pouch be filled on automated equipment? If so, is custom equipment required for filling?
Mohr: It is my understanding that the pouches can be filled on automated equipment; however, I’m sure engineering will be required.
Are the pouches supplied to brand owners pre-made and ready to fill?
Mohr: Yes, the pouches will be supplied pre-made.
Do you have any brand owners using this package commercially?
Mohr: This is a new packaging concept, and Bemis Healthcare Packaging does not currently have a commercial application.
______________________________________________________________________________________
See a host of new ideas in packaging machinery, materials and more for pharmaceuticals and other markets at EastPack 2016, June 14-16, in New York City.
______________________________________________________________________________________
About the Author(s)
You May Also Like




