Extended-text labels say a lot in a minimum of space
January 30, 2014

No packager wants anything more than making a product stand out in the marketplace. But sometimes, there are roadblocks to presenting a brand. For example, it can be a real challenge when the product has to be packaged in a small, ¾-in.-dia tube containing ½ oz. Placement of small packages in stores is equally challenging but vital to the retail sector, especially when it comes to point-of-sale merchandising. In point-of-sale, a brand's identity is like curb appeal in real estate. It can heighten awareness that leads to consideration and ultimately to product trial.
For skincare companies like Murad, Inc., El Segundo, CA, which offers Murad's a variety of skincare products in salons and cosmetics outlets, one roadblock is stringent federal regulations that require extensive information on packaging, including drug-fact panels when active ingredients are claimed. In order to comply, manufacturers typically package small tubes, bottles, and jars in an outer paperboard carton. Cosmetics and drugs that claim an active ingredient need to comply with regulatory requirements. So when Murad had an opportunity to promote two of its skincare products at point-of-sale through health and beauty retailer, Sephora, it wanted to make small, trial-size tubes stand tall on display, yet provide an ingredients statement, usage information and product facts. The tubes include Murad Acne Spot Treatment and Night Reform® Treatment products. Sephora planned to display the tubes in fishbowls, without cartons, positioned in a display at checkout counters.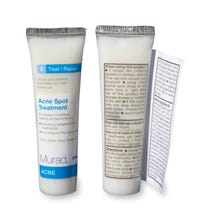
“Sephora specifically requested that the special Acne Spot Treatment and Night Reform promotion products not be packaged in a unit carton,” says Susie Becerra, Murad's purchasing manager. “The half-ounce tubes alone are better designed for impulse purchases than when contained in a carton. Unfortunately, there's very limited surface area [on their labels with which] to include the required information for regulatory compliance].” Each tube measures about 3 3/8 in. long, ±1/16in., and is ¾ in. dia.
Two-ply construction
Murad looked for a way to incororate the label information without compromising the overall visual appeal of the tube design and its brand identity, while still encouraging impulse purchases. It found what it was looking for in the FlexWrap™ extended-text label and the MultiVision® label line from WS Packaging Group (www.wspackaging.com). The patent-pending extended-text labels feature a durable, two-ply construction engineered for container diameters as small as 3/8 in.
Conformable to the clear, low-density polyethylene tubes with an extended applicator tip used for each treatment product, the label replaces a single-ply label. The FlexWrap's primary display panel base material is what WS Packaging describes as a 2.5-mil clear polyolefin, which helps maintain a no-label look, on a 1.2 clear Mylar® liner and an acrylic emulsion adhesive. The base label ply is topped with an additional ply of 2.5-mil ply of white polyolefin. The label copy is printed in four colors. Despite the small tube footprint (the labels provide an ample print area and measure 2 5/8 in. long by 2 5/8 in. wide), with which to educate consumers, the label can provide drug-fact panels, warnings and other details while incorporating mandatory information without extra over-packaging. Easy to open and reseal, the labels are applied with standard machiness in Murad's case by Contract Labeling Service (www.contractlabel.com) in Ontario, CA. The tubes are supplied by Berry Plastics (www.berryplastics.com). Berry also furnishes natural-toned glossy polypropylene caps.
“The result is an innovative label that provides the printing space for regulatory compliance and durability for long-term consumer use,” says Becerra. “The promotion was so successful, the tubes are now regular items for Sephora,” she adds.
More information is available: |
WS Packaging Group, 800/236-3424. www.wspackging.com. |
Berry Plastics Corp., 812/424-2904, berryplastics.com. |
Contract Labeling Service, Inc., 909/937-0344. www.contractlabel.com. |
About the Author(s)
You May Also Like


